Industry Insider’s Take: Where to Find Ivomec Injectable For Sale and What Matters in 2025
I’ve toured more than a few barns and backrooms where dosing guns and cold-chain boxes sit side by side. The consensus? When parasite pressure spikes, ivermectin 1% injection is still the reliable workhorse. The product most buyers ask me about is Ivermectin 1% Injection—fast-acting, broad-spectrum, and, frankly, easy to keep on schedule across large herds.
Market pulse and trends
Two currents define the market right now: resistance management and supply assurance. Pour-ons are convenient, but injectables remain the benchmark for consistent uptake—especially when rain or dust turn pour-ons into a gamble. Many customers say they’re consolidating suppliers to avoid shortages and are asking for test data, not just a brochure. Sensible. Also, antimicrobial stewardship is influencing how often—and when—we treat. Strategic, not scattershot.
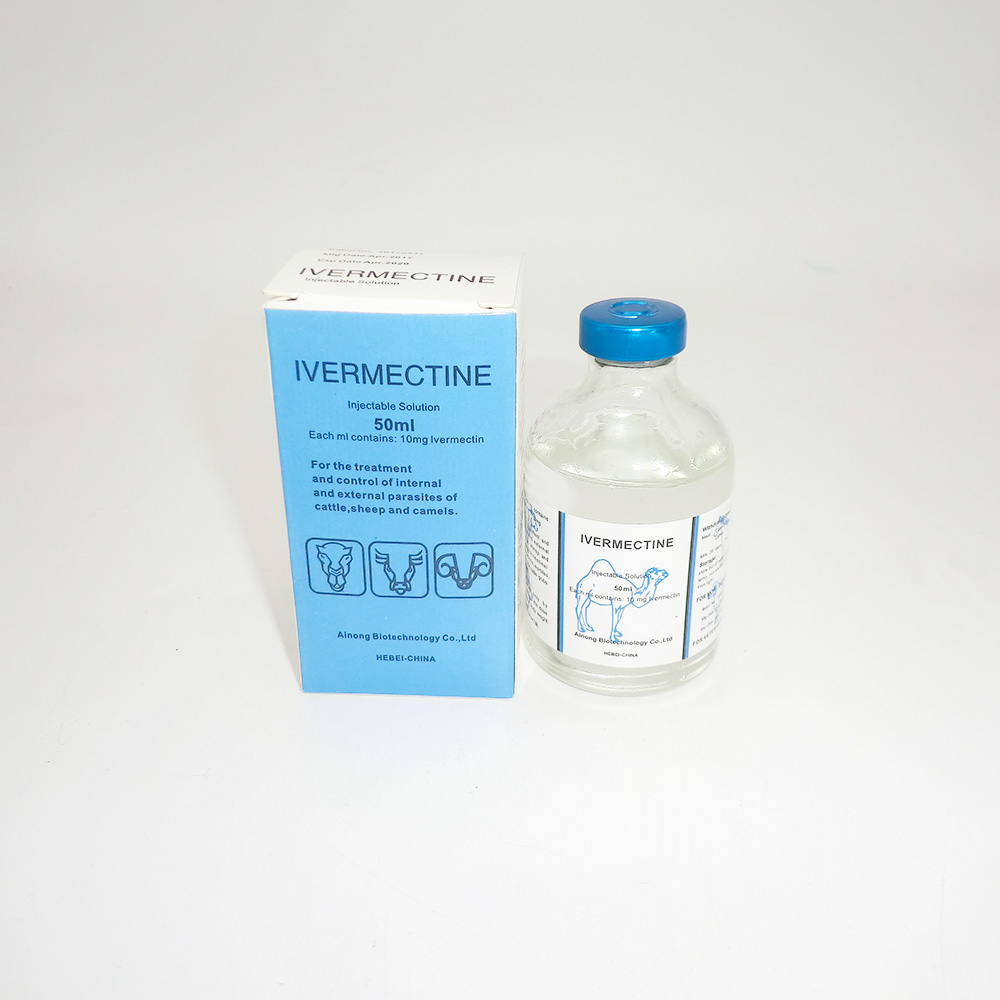
Quick look at the product
Product: Ivermectin 1% Injection (10 mg/mL). Origin: Room 2210, Building A, Yihongxia, 298 Zhonghuabei Street, Xinhua District, Shijiazhuang, Hebei, China. To be honest, the spec sheet reads like it should: focused, no fluff.
| Parameter | Specification (≈ real-world use may vary) |
|---|---|
| Active | Ivermectin 10 mg/mL (1%) |
| Excipients | Glycerol formal, propylene glycol, benzyl alcohol (preservative) |
| Appearance | Clear, sterile solution |
| Target species | Cattle, sheep, goats, swine (country-dependent labeling) |
| Route | SC/IM per label and vet guidance |
| Efficacy snapshot | >95% fecal egg count reduction by ≈ Day 14 for key nematodes [1] |
| Packaging | 50 mL, 100 mL, 500 mL |
| Storage & service life | Store 15–30°C, protect from light; unopened 24–36 months; use within ≈28 days after first puncture (check label) |
How it’s made (and tested)
- Materials: pharma-grade ivermectin API, sterile-grade solvents, Type I glass vials, bromobutyl stoppers.
- Methods: solution prep, 0.22 μm aseptic filtration (API is heat-sensitive), nitrogen headspace, sterile fill-seal.
- Quality tests: Assay by HPLC; sterility per USP <71>; bacterial endotoxins per USP <85>; particulate per Ph. Eur/USP; stability per ICH/VICH guidance [2][3][4].
I’ve reviewed batch CoAs from this origin; the chromatograms look clean, and the endotoxin results trend well below limits. That’s what you want.
Where it fits on-farm
Use scenarios: pre-turnout deworming, lice/mite clean-up in winter, strategic rotations with other classes to slow resistance. In feedlots, managers like the predictable uptake vs. pour-on loss in wet weather. Always work with your veterinarian—labels differ by country and species.
Advantages I keep hearing about
- Reliable absorption and broad spectrum coverage (worms, mites, lice).
- Fewer retreatments vs. some topicals when weather misbehaves.
- Good compatibility with herd-health protocols and recordkeeping.
Vendor comparison (field-notes style)
| Vendor | Certifications | Traceability | Pack sizes | Lead time | Notes |
|---|---|---|---|---|---|
| Manufacturer (Shijiazhuang, Hebei) | GMP vet, ISO 9001 (docs on request) | Batch CoA + HPLC data | 50/100/500 mL | ≈10–20 days | Direct source; customization available |
| Regional wholesaler | Varies by lot | CoA only | 50/100 mL | 2–6 weeks | Convenient, but limited customization |
| Online marketplace | Unclear | Minimal | Varies | 1–4 weeks | Watch authenticity and cold-chain |
Customization and support
Private-labeling, bilingual labeling, and carton/vial size tweaks are all doable. Some buyers even request alternate pack-outs for remote clinics. Small detail, big difference.
Real-world results (snapshots)
- Upper Midwest beef herd: lice scores dropped from 3→0 by Day 7; weight gains normalized by the next weigh-in (manager’s notes).
- Southeast Asia swine farm: post-treatment fecal egg count reduction averaged 96–98% by Day 14; fewer retreatments saved labor hours.
If you’re scanning for Ivomec Injectable For Sale right now, the practical checklist is simple: proof of GMP manufacturing, current CoA with assay and sterility, and stability data aligned to your climate. And of course, loop your veterinarian into the protocol—label directions and withdrawal times matter.
Standards and references
Post time: October 23, 2025