A pragmatic look at Pet Meds Without Prescription: what vets and buyers actually consider
Talking to clinic managers and shelter buyers lately, a theme keeps popping up: availability, compliance, and value. People search for Pet Meds Without Prescription when they’re under pressure to act fast. To be honest, that’s risky territory—antibiotics are tightly regulated for good reasons. Still, the market keeps evolving with better traceability, tougher testing, and clearer paperwork.
The product at the center of this conversation
One combo that industry folks ask me about: Tylosin Tartrate15mg + Doxycycline HCL10mg + Bromhexine HCL0.1mg tablet. It targets bacterial respiratory infections while Bromhexine helps clear mucus. Origin: Room 2210, Building A, Yihongxia, 298 Zhonghuabei Street, Xinhua District, Shijiazhuang, Hebei, China. In practice, clinics report quick relief from congestion and improved recovery—provided it’s used under veterinary oversight. Many customers say it seems fast-acting; I’d add, only when the bug profile matches.
Technical specifications (approximate, real-world use may vary)
| Actives (per tablet) | Tylosin Tartrate 15 mg; Doxycycline HCl 10 mg; Bromhexine HCl 0.1 mg |
| Dosage form | Compressed tablet, uncoated/film-coated options |
| Excipients (typ.) | Microcrystalline cellulose, lactose, povidone, croscarmellose, Mg stearate |
| Assay | ≥98% by HPLC (vendor data); content uniformity per USP/Ph. Eur. guidelines |
| Dissolution/Disintegration | USP / targets; verification via ISO 17025-accredited lab (on request) |
| Microbial limits | USP / compliant (batch COA) |
| Shelf life | ≈24 months sealed; stability per ICH Q1A (25°C/60% RH; 40°C/75% RH) |
| Packaging | HDPE bottle/blister; tamper-evident seal |
| Compliance | GMP facility (vendor reports), ISO 9001 QMS |
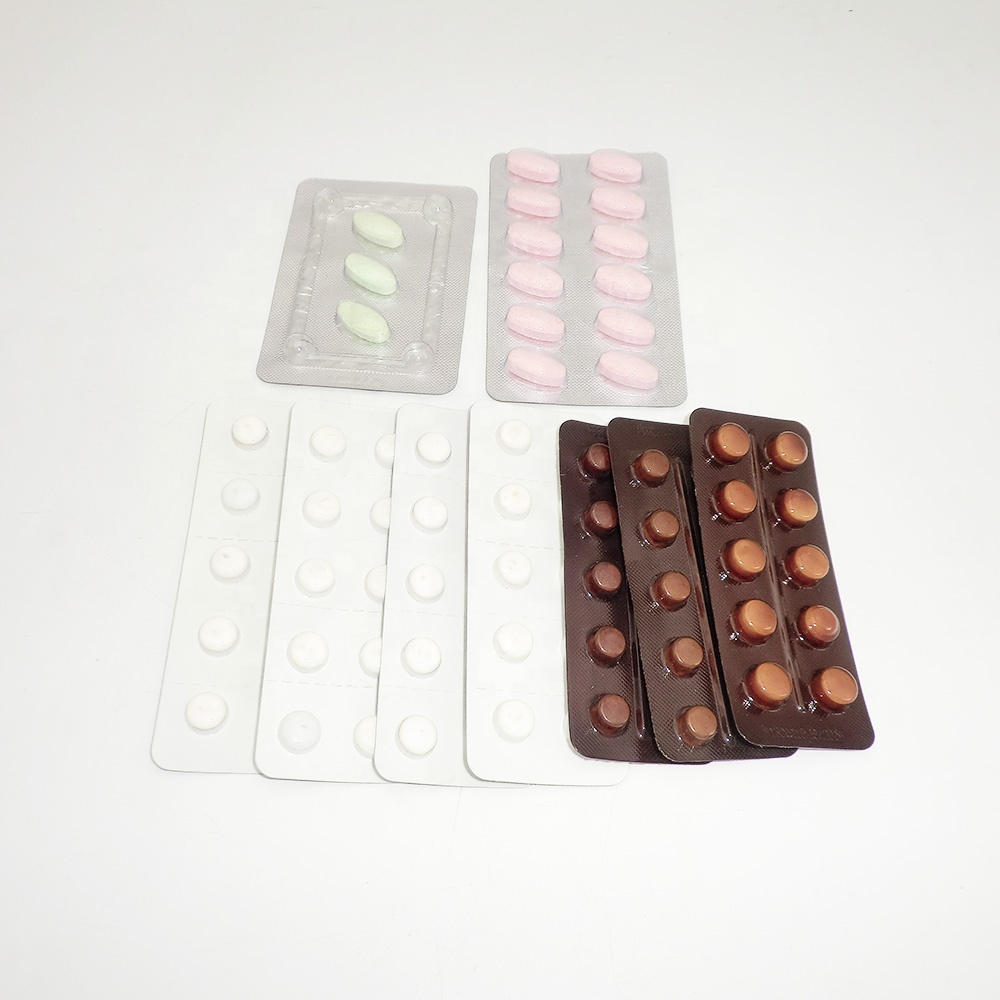
Process flow, methods, and QC
Materials are vetted via incoming QA (actives identity by FTIR/HPLC; excipient CoAs). The method is direct compression; blend uniformity is checked before tablet press. Finished batches undergo assay, dissolution, uniformity, hardness/friability, and microbial testing. Service life is validated with real-time and accelerated stability studies. Authorities increasingly expect antimicrobial stewardship documentation—no shortcuts here.
Application scenarios and industries
- Small animal clinics: bacterial respiratory infections with thick mucus.
- Shelters/rescues: outbreak control under veterinary protocols.
- Farm/kennel settings: targeted group treatments—only with vet direction.
Advantages noted: synergy of dual antibiotics with a mucolytic; easier breathing; fewer secondary visits when used appropriately. However, antimicrobial resistance is the elephant in the room—unsupervised use of Pet Meds Without Prescription can backfire.
Vendor comparison (indicative)
| Vendor | Quality & Docs | MOQ/Lead Time | Customization | 3rd-Party Tests | Price Tier |
|---|---|---|---|---|---|
| RC Pet (this product) | GMP, ISO 9001; full COA; stability summary | ≈10k tabs / 3–5 wks | Strength, coat, pack | Available on request | Mid |
| Generic importer A | Basic COA; limited traceability | ≈5k / 2–8 wks | Minimal | Seldom | Low |
| Clinic-only brand B | GMP, ISO; full dossier | N/A (Rx channel) | Limited | Standard | High |
Customization and documentation
Buyers typically request private labeling, altered strengths, and blister vs. bottle. The smarter ones also insist on batch-specific COAs, method validation summaries, and a QP or responsible person sign-off. If you’re tempted by Pet Meds Without Prescription, at least secure GMP proof and independent assays.
Case study (condensed)
A mid-size rescue reported a kennel cough cluster. Under a vet protocol, they deployed this combo for clinically indicated cases; mucus clearance improved within days, and new cases dropped by week two. What mattered: culture results informed therapy, and they tracked outcomes. No DIY shortcuts, no blanket use.
Regulatory note
Antibiotics are prescription-only in many jurisdictions. Always consult a licensed veterinarian; comply with FDA CVM/EMA/VMD guidance on antimicrobial stewardship. Self-medicating animals with Pet Meds Without Prescription isn’t just risky—it can fuel resistance.
References
- FDA Center for Veterinary Medicine. Antimicrobial Stewardship in Veterinary Settings. https://www.fda.gov/animal-veterinary/safety-health/antimicrobial-resistance
- EMA/CVMP. Reflection paper on antimicrobials in animals. https://www.ema.europa.eu
- WOAH (OIE). Responsible and Prudent Use of Antimicrobial Agents in Veterinary Medicine. https://www.woah.org
- USP General Chapters , , , ; ICH Q1A(R2) Stability Testing. https://www.usp.org; https://www.ich.org
Post time: October 27, 2025